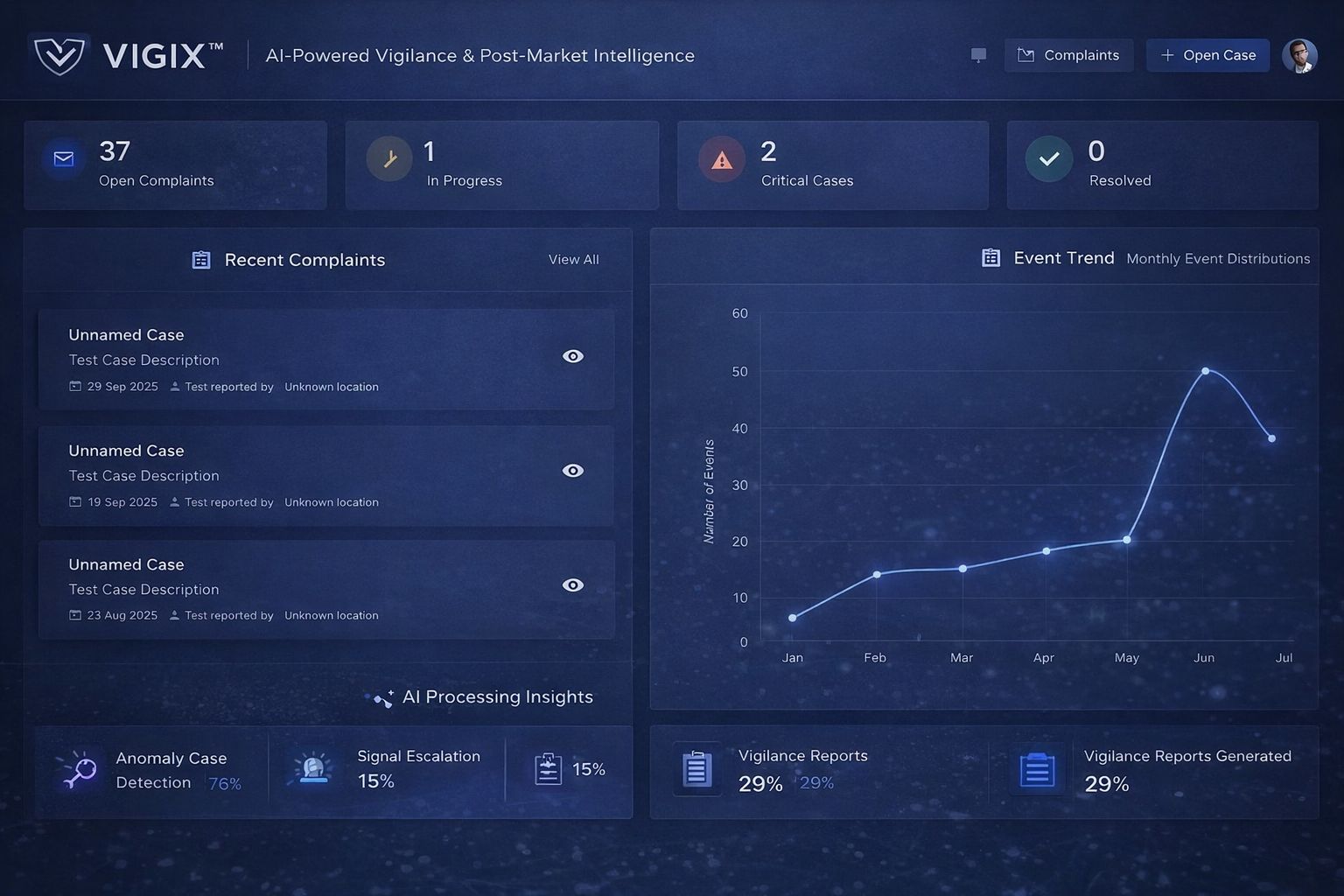
VigiX 360 — Bioexcel’s Integrated Medical Device Compliance & Evidence Platform
Unifying Clinical Investigations, Clinical Evaluation, Real-World Evidence, Materiovigilance, and PMS reporting into one structured,audit-ready system.
Designed for manufacturers operating under EU MDR, ISO 14155, ISO 13485, FDA, CDSCO, and global vigilance frameworks.
WHY VIGIX 360
- Clinical investigations in one system
- CER in documents
- PMCF data in spreadsheets
- Complaints in ticket tools
- PMS reports manually compiled
- Risk and CAPA disconnected
This creates regulatory risk and operational inefficiency.
VigiX 360 connects the entire evidence lifecycle.
CORE DESIGN PRINCIPLE
One Shared Core Platform + Switchable Modules
- Modular activation per
- Multi-product scalability
- Region-specific rule configuration
- Full lifecycle traceability
This ensures compliance - PMS reports manually compiled
- Risk and CAPA disconnected
This ensures compliance maturity without operational chaos.
PLATFORM MODULES (UPDATED – INCLUDING RWE / PMCF)
RWE/PMCF Module
(Real-World Evidence & Post-Market Clinical Follow-Up)
This module transforms VigiX 360 into a continuous evidence intelligence system.
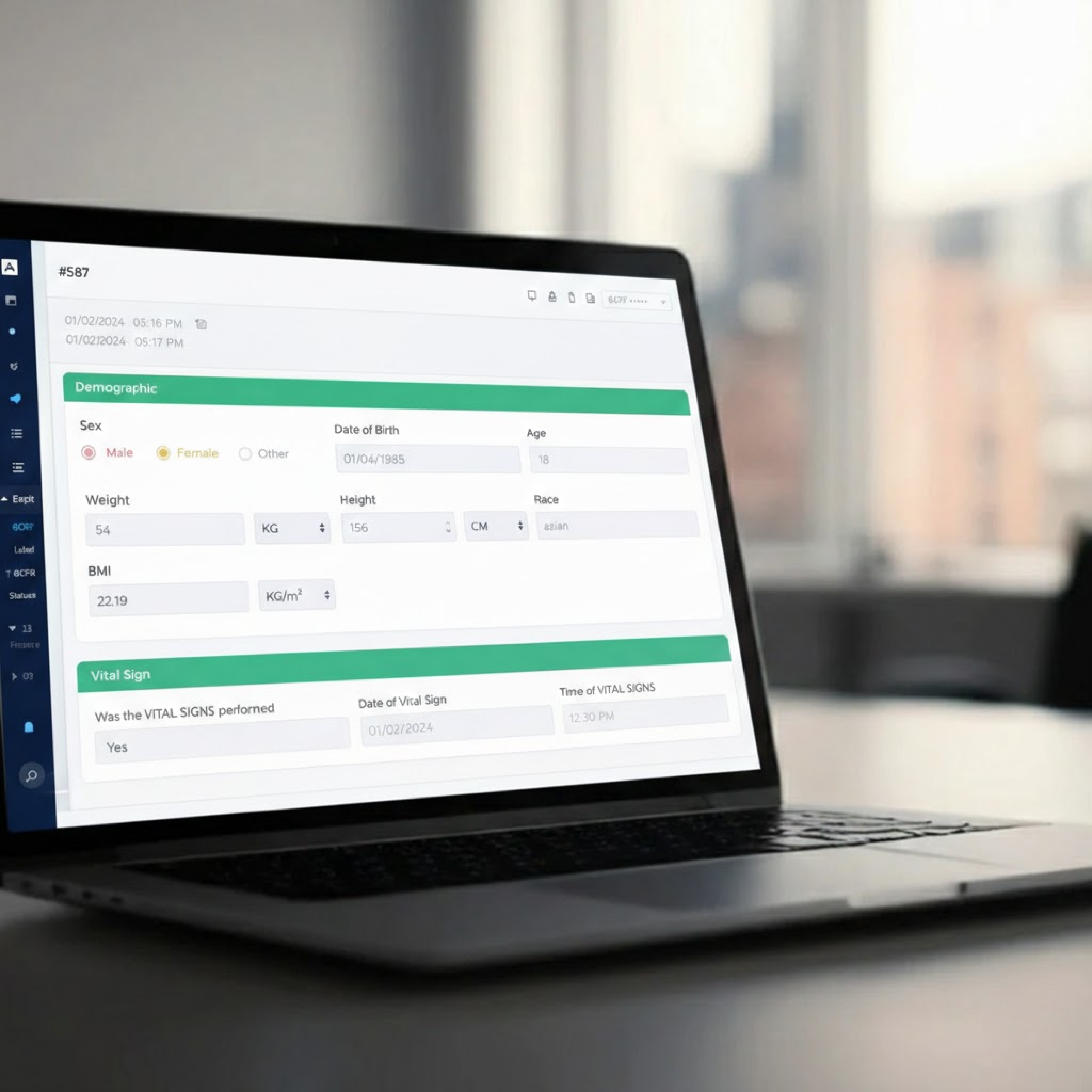
What is RWE in VigiX 360?
Real-World Evidence (RWE) captures device performance and safety data from real-world use environments.
Typically sourced from:
- PMCF studies
- Surveys
- Registries
- Observational follow-ups
- Hospital datasets
- Complaint & servicing data
- Published real-world literature

PMCF Management Capabilities
VigiX 360 structures:
- PMCF Planning workflows
- PMCF Study tracking
- Survey creation & tracking
- Follow-up documentation
- Data collection oversight
- Registry tracking
Outputs include:
- PMCF Plan
- PMCF Evaluation Report
- Structured evidence summaries

Complaint & Servicing Data as Evidence
VigiX 360 allows:
- Linking complaint data to RWE dashboards
- Linking service records to safety trends
- Structured device performance tracking
This converts routine post-market data into usable clinical evidence.
Published RWE Integration
- Literature tracking for real-world studies
- Structured evaluation & linkage to CER
- Evidence package creation








