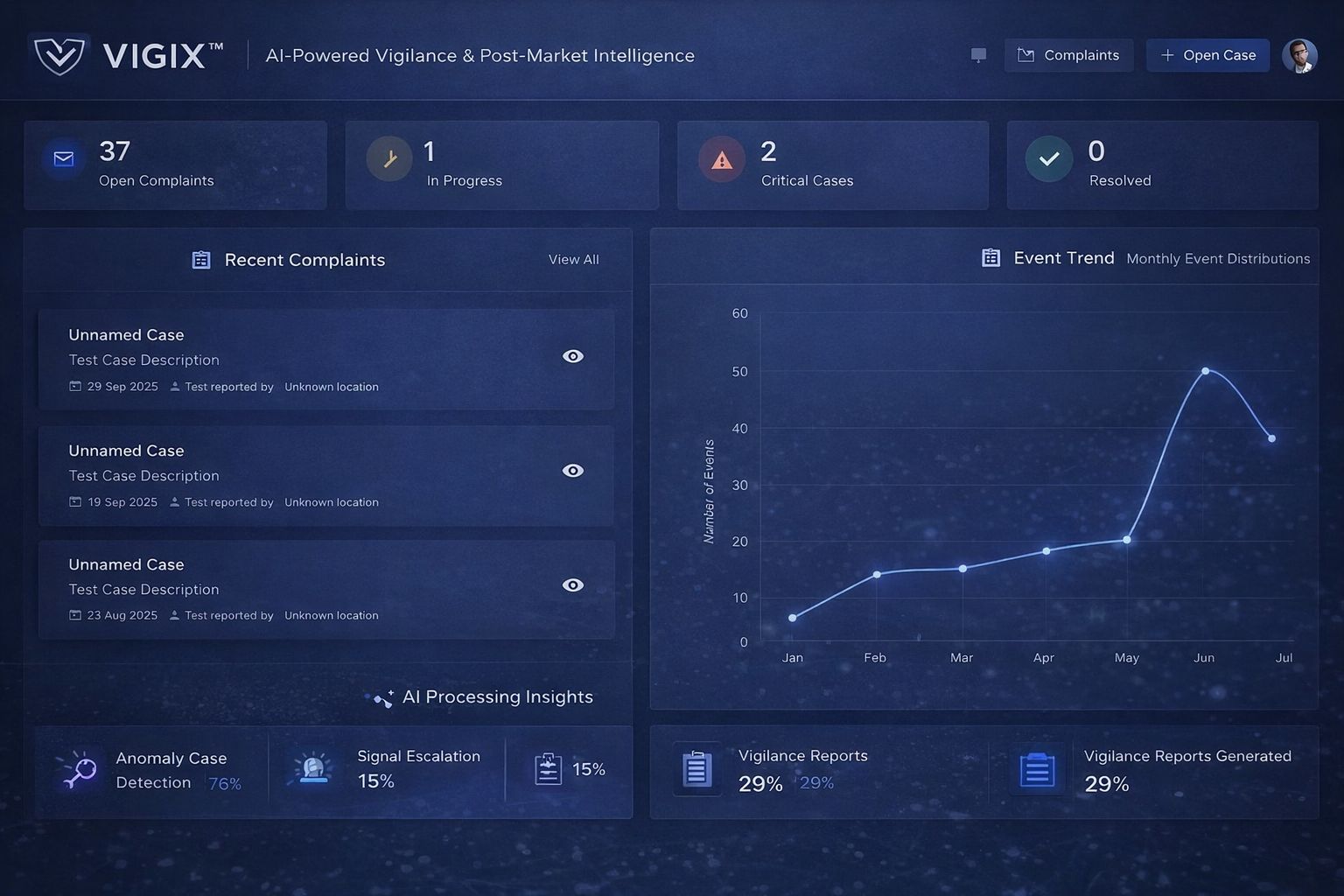
Vigi X™ – Materiovigilance & Post‑Market Compliance Platform
Vigi X™ is Bioexcel’s digital materiovigilance and post‑market compliance platform, designed to support manufacturers in meeting EU MDR post‑market surveillance and vigilance obligations.
The platform enables structured handling of complaints, incidents, PMCF data, and post‑market reports while maintaining regulatory traceability.
Capabilities
- Complaint intake and classification
- Incident assessment and reportability decision support
- PMS data aggregation and trending
- PMCF data integration
- PMSR and PSUR support
- Regulatory reporting workflow support (EU / UK / US / India)
Regulatory
- EU MDR Articles 83–86 (PMS)
- ISO 13485 post‑market processes
- EU MDR vigilance requirements

Frequently Asked Questions (FAQ)
ANSWER – ISO 10993 biological evaluation is applied based on
device contact type and duration. The evaluation approach is justified through a biological evaluation plan.
Answer-Bioexcel supports planning, interpretation, and
documentation. Testing is performed by qualified laboratories; we support selecting appropriate tests and
presenting results.
ANSWER – A TRA evaluates the toxicological
risk of substances identified from materials and chemical characterization, supporting safety justification.
ANSWER – Yes. Toxicology and biocompatibility documentation forms part of the technical documentation and supports NB review.


